How to Prepare Human Kidney Organoids
The kidney is one of the vital organs in the human body, responsible for functions such as metabolite waste removal and maintaining electrolyte balance, ensuring the stability of the body's internal environment. Kidney organoids are 3D models of kidney function, beneficial for modeling human kidney diseases. They are mostly induced from human pluripotent stem cells (PSCs) in vitro and can express various structures similar to human renal units, including podocytes, proximal tubules, distal tubules, and collecting ducts.
Kidney Organoid Culturing Protocol
We have compiled the culturing protocol for generating human kidney organoids from human pluripotent stem cells based on the article published in Nat Protoc.[1].
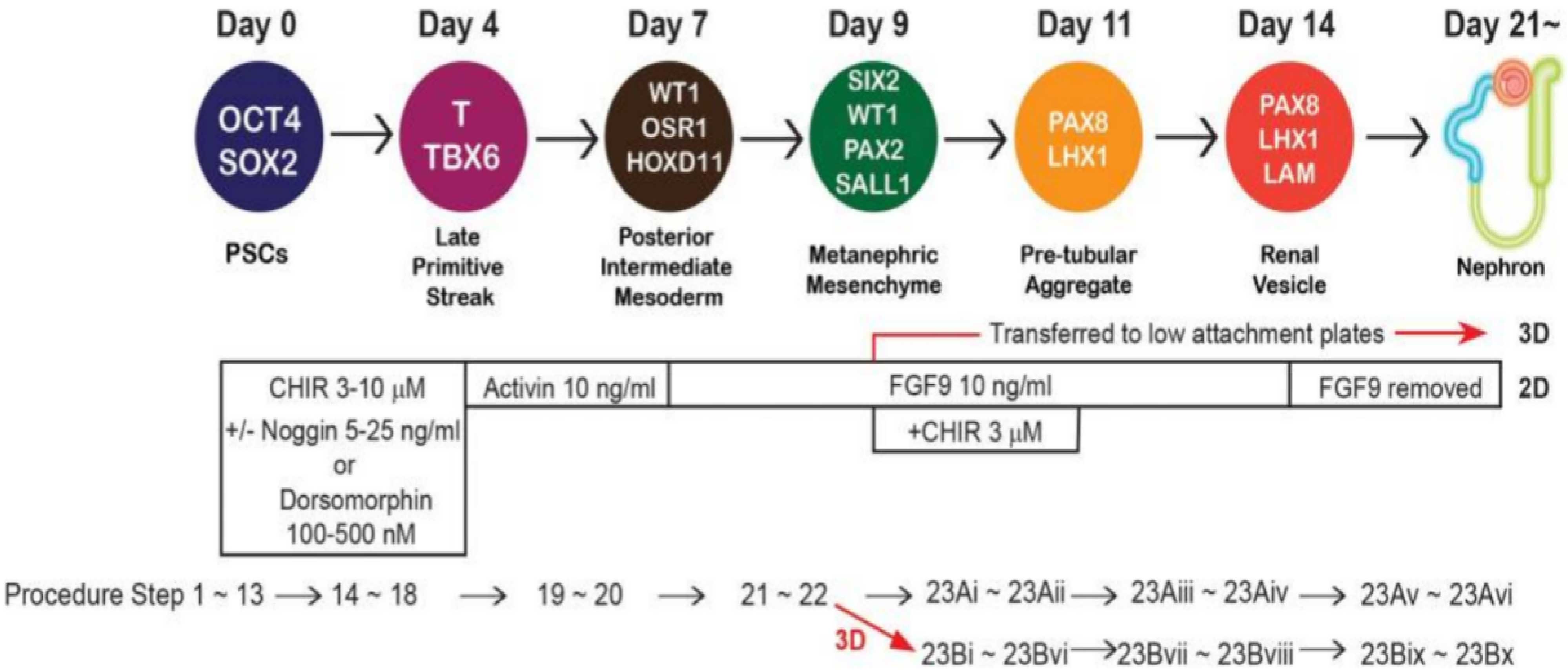
Figure 1. Process of hPSC Differentiation into Kidney Organoids (PMID: 28005067)[1]
1. Human pluripotent stem cell (hPSC) culture
hPSCs are seeded onto a 6-well plate coated with LDEV-free extracellular matrix substrate and cultured in ReproFF2 medium (containing 10 ng/mL FGF2). Healthy undifferentiated hPSCs exhibit well-defined round colony morphology, and they are passaged every seven days using dissociation solution for iPSCs.
2. Differentiation of human pluripotent stem cells
When cell density reaches 80%, cells are dissociated using Accutase, resuspended in ReproFF2 medium (containing 10 μM Y27632), and seeded onto a 24-well plate coated with LDEV-free extracellular matrix substrate. Cells are cultured for 3 days and then switched to Advanced RPMI 1640 medium (containing 1× L-GlutaMAX, 3~10 μM CHIR99021, 5-25 ng/mL Noggin, 100-500 nM Dorsomorphin) for continued culture with media changes every two days until cells form loosely packed dense cell clusters (usually within 96 h). Subsequently, the medium is changed to Advanced RPMI 1640 (containing 1× L-GlutaMAX, 10 ng/mL activin A) and cultured for an additional 2-3 days.
3. Induction of hPSC differentiation into neural progenitor cells (NPCs)
The medium is changed to Advanced RPMI 1640 (containing 1× L-GlutaMAX, 10 ng/mL FGF9) and cultured for 2 days.
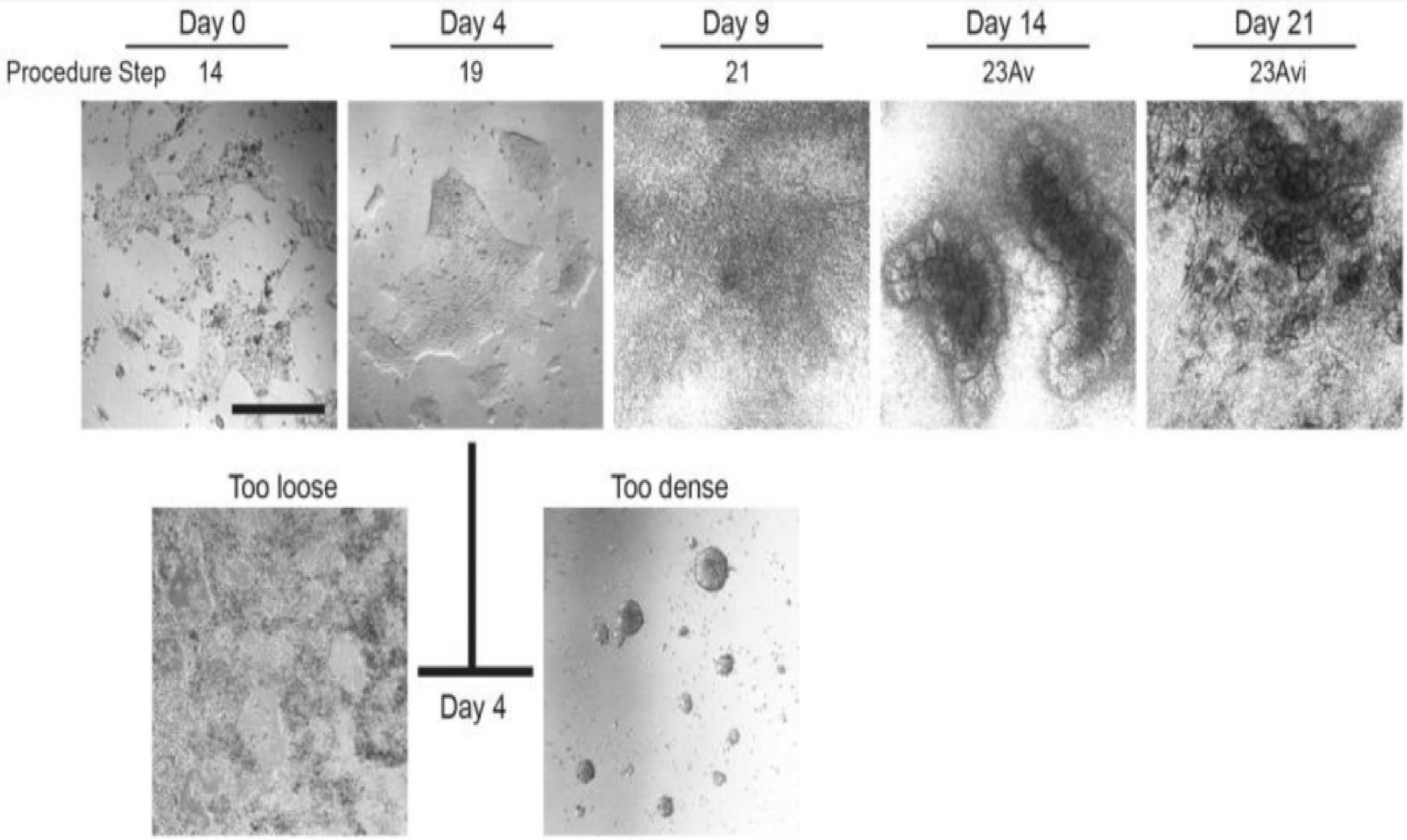
Figure 2. Morphological changes during hPSC differentiation.
Day 0:Undifferentiated hPSCs (H9) at the start of differentiation;Day 4: Late primitive streak stage;Day 9: Nephron progenitor cell stage;Day 14: Renal vesicle stage;Day 21: Renal unit stage;Optimal morphology of cells treated with Activin A on day 4 is characterized by loosely dense clusters.
4. Organoid induction
4.1 Differentiation of NPCs into kidney organoids (2D)
After step 3, switch to Advanced RPMI 1640 medium (containing 1× L-GlutaMAX, 10 ng/mL FGF9, 3 μM CHIR99021) and continue incubation for 2 days. Then switch to Advanced RPMI 1640 medium (containing 1× L-GlutaMAX, 10 ng/mL FGF9) and culture for 2-3 days. If polarized structures resembling renal vesicles with lumens are observed, proceed to the next step. If renal vesicle structures are not observed within 3 days, confirm the expression of LHX1 through immunostaining. Then switch to Advanced RPMI 1640 medium (containing 1× L-GlutaMAX) and continue culture, changing the medium every 2-3 days. Kidney organoids remain stable throughout at least 3 months of differentiation, and the renal unit structure can be identified under a microscope.
4.2 Differentiation of hPSCs into kidney organoids (3D)
After step 2, digest the cells using Accutase and resuspend them in Advanced RPMI 1640 medium (containing 1× L-GlutaMAX, 10 ng/mL FGF9, 3 μM CHIR99021). Seed the cells into ultra-low attachment round-bottom 96-well plates and culture for 2 days. Then switch to Advanced RPMI 1640 medium (containing 1× L-GlutaMAX, 10 ng/mL FGF9) and continue culture for 2-3 days until renal vesicle-like spherical structures are observed under a microscope.
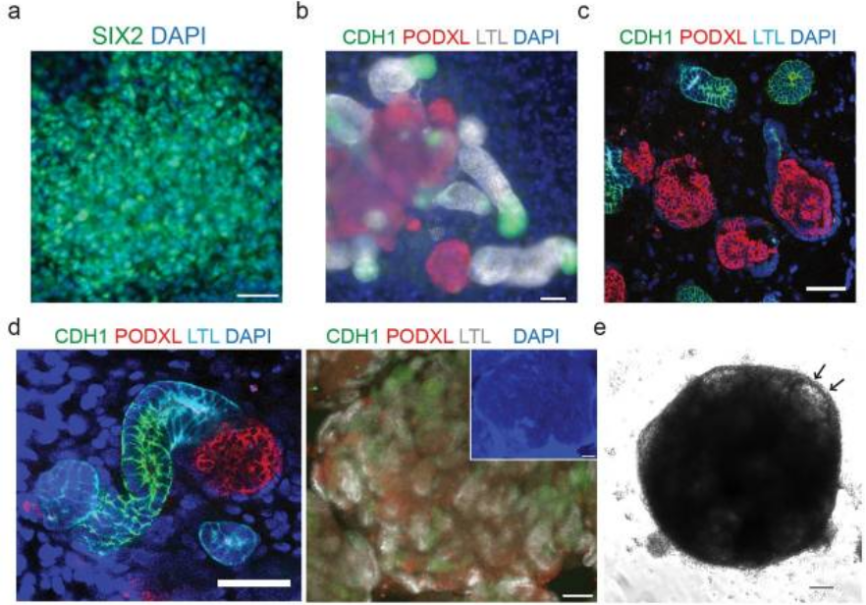
Figure 3. Immunostaining of NPC and renal units (PMID: 28005067)[1]
(a) Immunofluorescence staining of SIX2 on day 9 of differentiation shows NPC. Scale bar: 50 μm; (b) Immunofluorescence staining to identify differentiated renal unit structures on day 21 of 2D culture. Scale bar: 50 μm; (c) Identification of renal unit structures on day 21 of differentiation in 3D culture using frozen sections. Scale bar: 50 μm; (d) Immunostaining of renal units on day 28 (left, scale bar: 50 μm) and day 21 (right, scale bar: 100 μm) in 3D culture. PODXL: a podocyte marker, LTL: a proximal tubule marker, CDH1: Henle's loop and distal tubule marker; (e) Bright-field image of organoids on day 21 in 3D culture, with arrows indicating glomerular structures.
Related products
|
Product Type |
Product Name |
Catalog Number |
Specification |
|
Recombinant Protein |
Recombinant Human Noggin Protein,His Tag |
C230462 |
5 μg/100 μg/500 μg |
|
Recombinant Protein |
Recombinant Human/Mouse/Rat Activin A Protein |
C230520 |
10 μg/100 μg/500 μg |
|
Recombinant Protein |
Recombinant Human FGF-9 Protein, His tag |
C230347 |
5 μg/100 μg/500 μg |
|
Recombinant Protein |
Recombinant Human FGF-10 Protein, His Tag |
C230418 |
5 μg/100 μg/500 μg |
|
Recombinant Protein |
Recombinant Human bFGF/FGF-2 Protein |
C230295 |
10 μg/100 μg/500 μg |
|
Cell Dissociation Solution |
Accutase |
C230116 |
100 mL |
